Natural Product Synthetic Biology
Reprogramming the chemistry of life
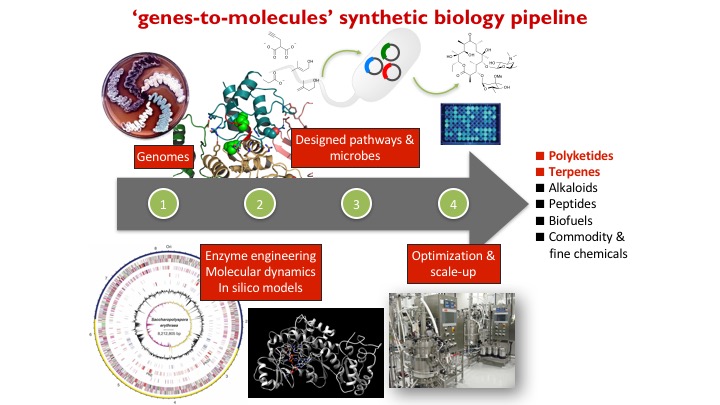
We are developing a genes-to-molecules synthetic biology pipeline for the synthesis, diversification, and discovery of natural products for drug discovery. We are interested in (1) how natural products are biosynthesized in Nature, (2) how to reprogram natural product biosynthesis to make new analogues, and (3) structure-function relationship studies of biologically active natural products.